Direct electrochemical splitting of liquid ammonia for on-site hydrogen production at ambient temperature
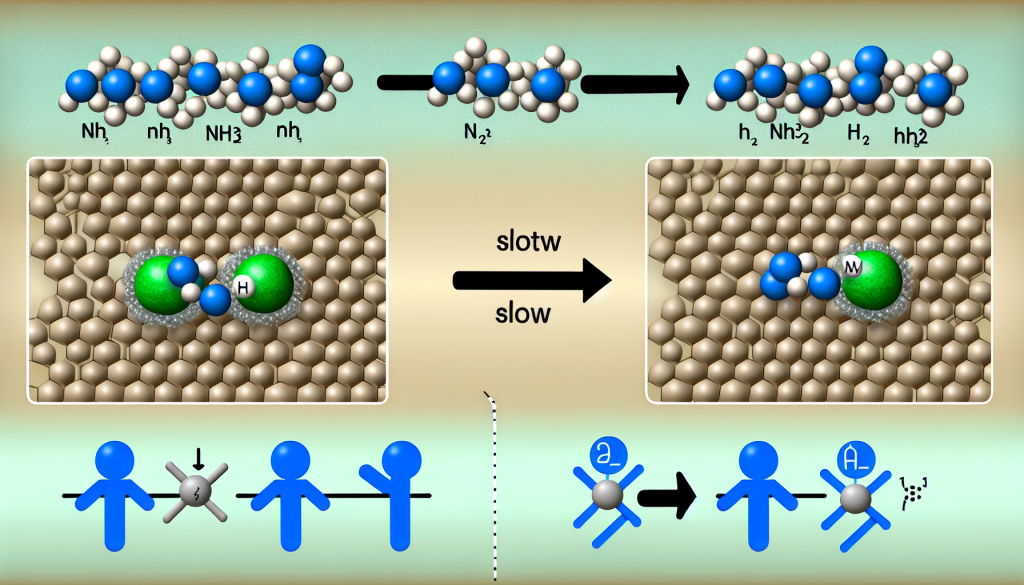
Direct electrochemical splitting of liquid ammonia enables efficient on-site hydrogen production at room temperature and low pressure (<1 MPa) by employing ruthenium nanoparticles anchored on nitrogen-doped carbon. This advance bypasses the high-temperature limitations of conventional ammonia cracking, introducing a more sustainable route to hydrogen generation with excellent catalytic activity and stable operation exceeding 100 hours, outperforming commercial Pt/C systems.
Credit: Chinese Journal of Catalysis
Ensuring safe and effective hydrogen storage and transport remains a central challenge in building a sustainable hydrogen economy. Ammonia stands out as a practical hydrogen carrier due to its high hydrogen content and ease of liquefaction. However, converting ammonia back into hydrogen traditionally demands elevated temperatures between 400 and 700 °C. Electrochemical liquid ammonia decomposition (ELADH) provides an appealing alternative, enabling hydrogen release at ambient temperature with a relatively low theoretical cell voltage. Despite its promise, progress in this field has been constrained by slow reaction kinetics, limited catalyst durability, and the harsh, corrosive nature of the reaction environment.
A recent study led by Prof. Jun-Min Yan of Jilin University and Prof. Hai-Xia Zhong of the Changchun Institute of Applied Chemistry, Chinese Academy of Sciences, reports a highly active and durable ELADH system. Using density functional theory calculations, the researchers determined that ruthenium outperforms platinum, rhodium, and iridium as a catalyst, with the Ru (101) crystal facet offering a low energy barrier for N–H bond cleavage and favorable hydrogen adsorption characteristics. Based on these insights, they prepared nitrogen-doped carbon-supported ruthenium nanoparticle catalysts (Ru NPs-CN) through a two-step pyrolysis strategy.
The resulting Ru NPs-CN catalyst, characterized by predominantly exposed (101) facets and a porous architecture, demonstrated exceptional performance in an optimized electrochemical setup featuring a graphite plate anode and NH4PF6 electrolyte in liquid ammonia. The system achieved a high current density of −910 mA cm−2 at −1.47 V and required an overpotential of only −1.01 V to reach −10 mA cm−2, markedly surpassing both ruthenium single-atom catalysts and commercial Pt/C. Notably, stable hydrogen production was maintained for more than 100 hours in a two-electrode configuration, highlighting its impressive operational durability. This research establishes a practical pathway for hydrogen generation from ammonia under mild conditions and advances fundamental understanding of electrochemical ammonia splitting. The findings were published in Chinese Journal of Catalysis (DOI: 10.1016/S1872-2067(25)64915-1).
About the journal
Chinese Journal of Catalysis is jointly sponsored by the Dalian Institute of Chemical Physics, Chinese Academy of Sciences, and the Chinese Chemical Society, and is published by Elsevier. Issued monthly in English, the journal features rigorously peer-reviewed contributions spanning all aspects of catalysis research. It includes Reviews, Accounts, Communications, Full Articles, Highlights, Perspectives, and Viewpoints that address both fundamental principles and practical applications. The journal is ranked among the leading publications in Applied Chemistry and currently holds an SCI impact factor of 17.7.
Journal website: Chinese Journal of Catalysis
Manuscript submission: Chinese Journal of Catalysis online submission system
Journal
Chinese Journal of Catalysis
DOI
10.1016/S1872-2067(25)64915-1
Article Title
Direct electrochemical liquid ammonia splitting for onsite hydrogen generation under room temperature
Article Publication Date
3-Feb-2026
